 
The colliding electron must have kinetic energy greater than or equal to the difference between energy levels of the atom. In the case of an emission spectrum, the atom is first excited by a colliding electron. Hence, the parts of the spectrum corresponding to these wavelengths appear dark by comparison with the other wavelengths not absorbed.ĭistinguish between emission and absorption line spectra The atoms of the gas absorb light of the same wavelengths which they can emit, and then re-radiate the same wavelengths almost immediately but in all directions.White light is passed through the tube.Gases such as hydrogen are placed in a tube.Steps to obtain absorption line spectrum: As these frequencies of light are now missing, they account for the dark lines in the absorption spectrum, which is discrete dark line on a continuous spectrum Since the energy levels are discrete, only photons of certain frequencies are absorbed. Those incident photons whose energies are exactly equal to the difference between the atom’s energy levels are being absorbed. Hence, there will be no separated/isolated lines of definite frequency. 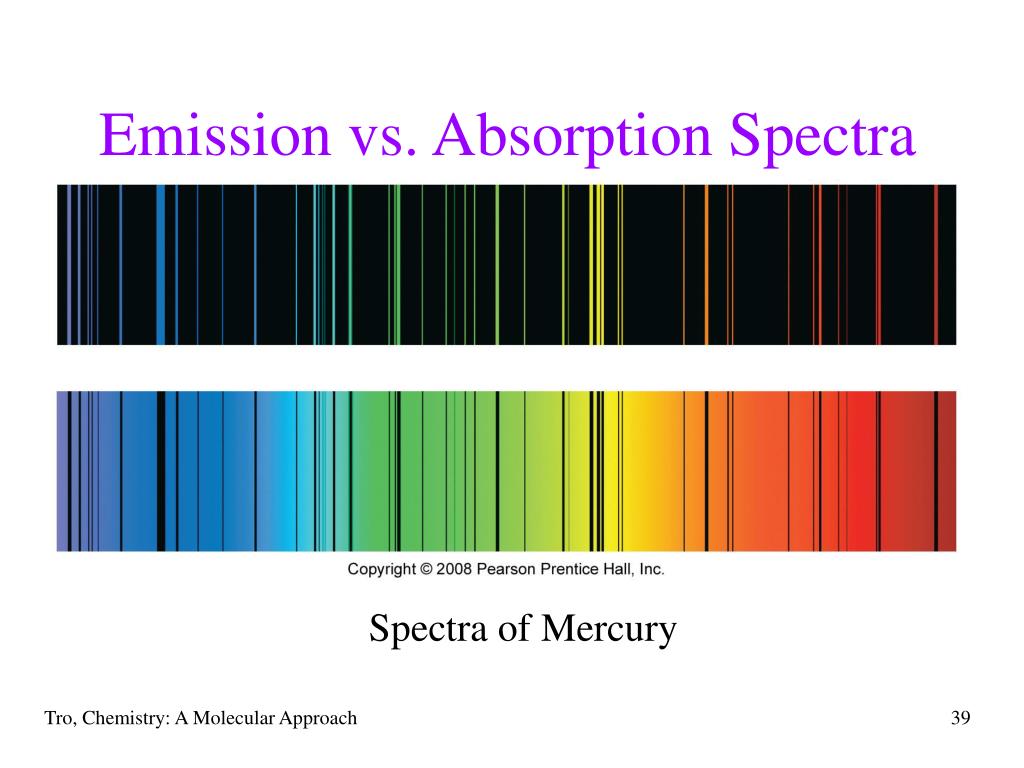
At high pressure, tightly packed gas atoms or molecules will be vibrating, rotating or colliding with each other, such that many more energy levels will be created. If the gases used are not at low pressure, there will be a continuous range of colours. No two gases give the same exact line spectrum. The well-defined separation of lines is experimental evidence for the existance of separate or ‘quantized’ energy levels in the atom. The emitted light are analyzed with a spectrometer and discrete bright lines in a dark background are observed. Only certain frequency lines are present in the spectrum as only certain high to low energy level transitions are possible within the atom. 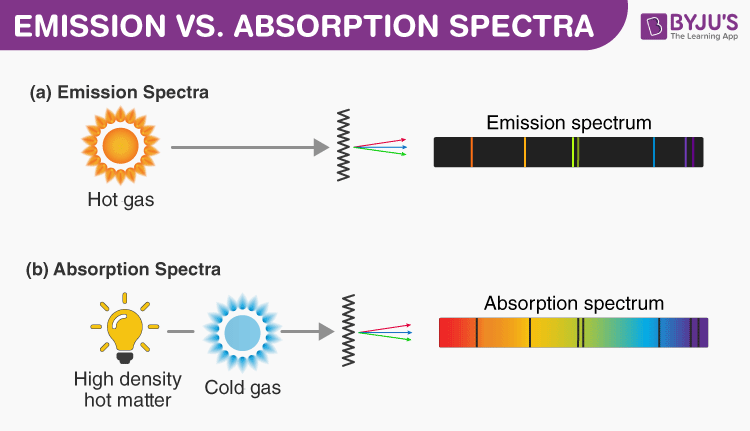
The frequency f of the emission line is dependent on the difference between the high and low energy levels.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
